What Is A Titration And How Is It Performed

Titration Experiments In Chemistry The Chemistry Blog Swirl the water around a few times before opening the stopcock and allowing it to drain. [2] repeat the rinsing process at least 3 times with water to completely clean your burette. after you’ve rinsed the burette with water, perform the same rinsing process at least 2 times with your analyte. 3. Titration. burette, laboratory apparatus used in quantitative chemical analysis to measure the volume of a liquid or a gas. it consists of a graduated glass tube with a stopcock (turning plug, or spigot) at one end. on a liquid burette, the stopcock is at the bottom, and the precise volume of the liquid dispensed can be determined by reading.
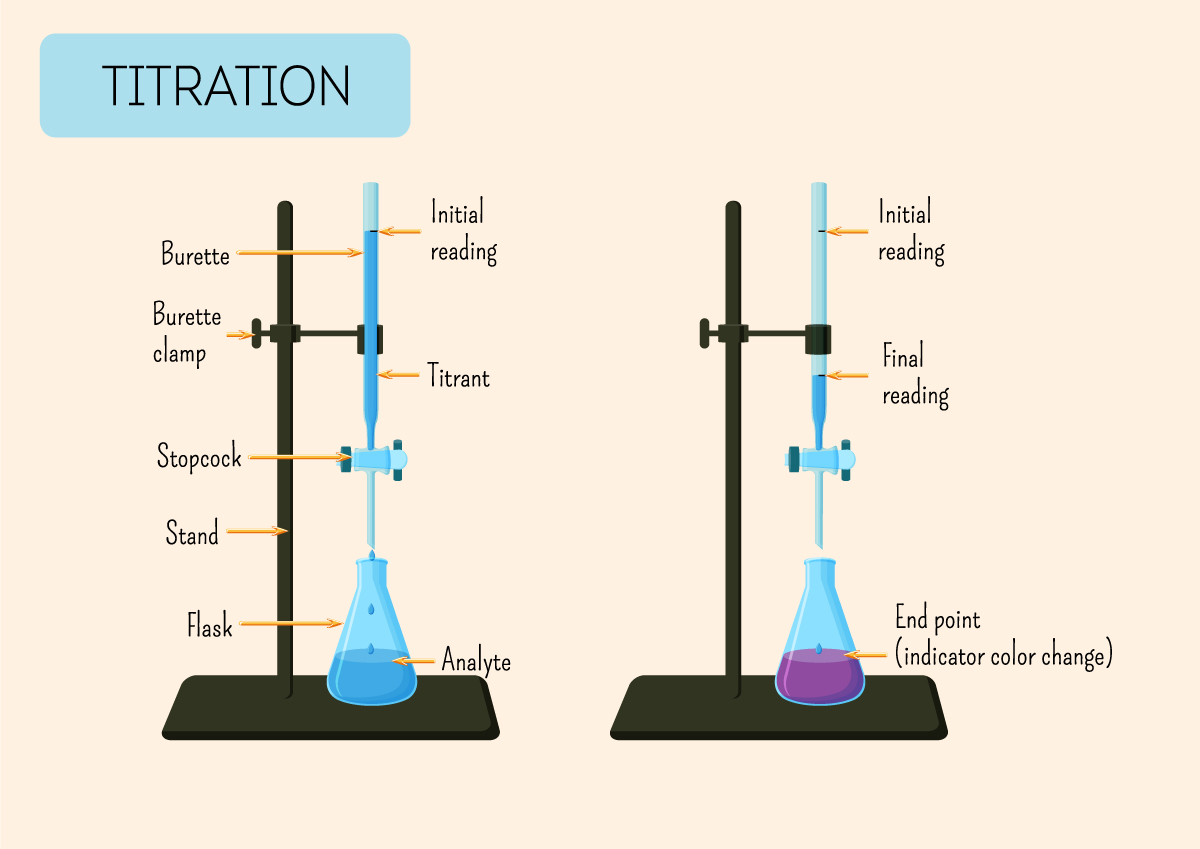
Chemistry How To Titration The Edge Titration is a way of measuring the concentration of something, usually the concentration of a substance in a solution. for example, you can use it to find the concentration of a solution of an acid. titration is a type of chemical analysis. we consider it to be analysis because we use it to make a measurement. Titration is the slow addition of one solution of a known concentration (called a titrant) to a known volume of another solution of unknown concentration until the reaction reaches neutralization, which is often indicated by a color change. the solution called the titrant must satisfy the necessary requirements to be a primary or secondary. An assay is a type of biological titration used to determine the concentration of a virus or bacterium. serial dilutions are performed on a sample in a fixed ratio (such as 1:1, 1:2, 1:4, 1:8, etc.) until the last dilution does not give a positive test for the presence of the virus. A titration is a laboratory technique used to precisely measure molar concentration of an unknown solution using a known solution. the basic process involves adding a standard solution of one reagent to a known amount of the unknown solution of a different reagent. for instance, you might add a standard base solution to an mystery acid solution.

How To Do Titrations In Chemistry An assay is a type of biological titration used to determine the concentration of a virus or bacterium. serial dilutions are performed on a sample in a fixed ratio (such as 1:1, 1:2, 1:4, 1:8, etc.) until the last dilution does not give a positive test for the presence of the virus. A titration is a laboratory technique used to precisely measure molar concentration of an unknown solution using a known solution. the basic process involves adding a standard solution of one reagent to a known amount of the unknown solution of a different reagent. for instance, you might add a standard base solution to an mystery acid solution. A titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to determine its concentration. many titrations are acid base neutralization reactions, though other types of titrations can also be performed. How a titration is performed . a typical titration is set up with an erlenmeyer flask or beaker containing a precisely known volume of analyte (unknown concentration) and a color change indicator. a pipette or burette containing a known concentration of titrant is placed above the flask or beaker of an analyte.

What Is Titration And How Is It Done Chemistry Made Simple A titration is an experiment where a volume of a solution of known concentration is added to a volume of another solution in order to determine its concentration. many titrations are acid base neutralization reactions, though other types of titrations can also be performed. How a titration is performed . a typical titration is set up with an erlenmeyer flask or beaker containing a precisely known volume of analyte (unknown concentration) and a color change indicator. a pipette or burette containing a known concentration of titrant is placed above the flask or beaker of an analyte.

How To Do Titrations In Chemistry

Comments are closed.